Recently, Signal Transduction and Targeted Therapy (STTT) (SCI IF=52.7), a leading international journal under the Nature portfolio, cited the complete clinical research series of China's first independently developed oral anti-HIV drug - Aidea Pharma's (stock code: 688488.SH) Ainuovirine tablets and its fixed-dose combination Ainuomiti tablets (Aitribond®)—in an article titled "Current Landscape of Innovative Drug Development and Regulatory Support in China"[1].
The article specifically points out: “Ainuovirine and its fixed-dose combination represent China's first self-developed novel antiretroviral combination therapy, offering people living with HIV (PWH) more treatment options. These drugs have attracted significant attention for their breakthrough role in addressing critical public health needs and were granted accelerated market approval through the National Medical Products Administration's priority review pathway. This has enabled faster access to these urgently needed therapies for Chinese PWH, improving drug availability.”

Clinical Evidence: Dual Assurance of Metabolic Safety and Sustained Viral Suppression
Throughout the clinical development of Ainuovirine tablets and Aitribond®, multiple rigorous studies have consistently demonstrated their dual advantages in metabolic safety and sustained viral suppression:
The 48-week data from the RACER study[2] showed that the incidence of neuropsychiatric adverse events in the Ainuovirine group (17.5%) was significantly lower than that in the Efavirenz group (54.1%), fully demonstrating Ainuovirine's excellent neuro-safety profile in long-term treatment.
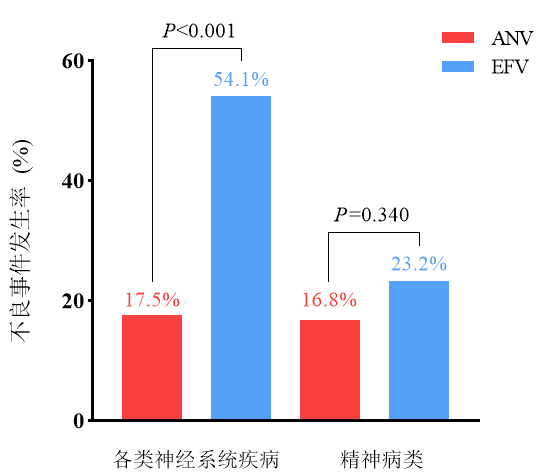
Systemic Diseases with >5% Difference in Adverse Event Incidence
At 48 weeks, the SPRINT study results[3] showed that the Aitribond® group performed better than the control group in both the incidence and severity of dyslipidemia-related adverse events (classified according to MedDRA). Not only was the overall reporting rate lower, but the proportion of moderate-to-severe events was also significantly reduced, demonstrating its comprehensive health benefits in improving lipid profiles and reducing long-term cardiovascular and metabolic risks.
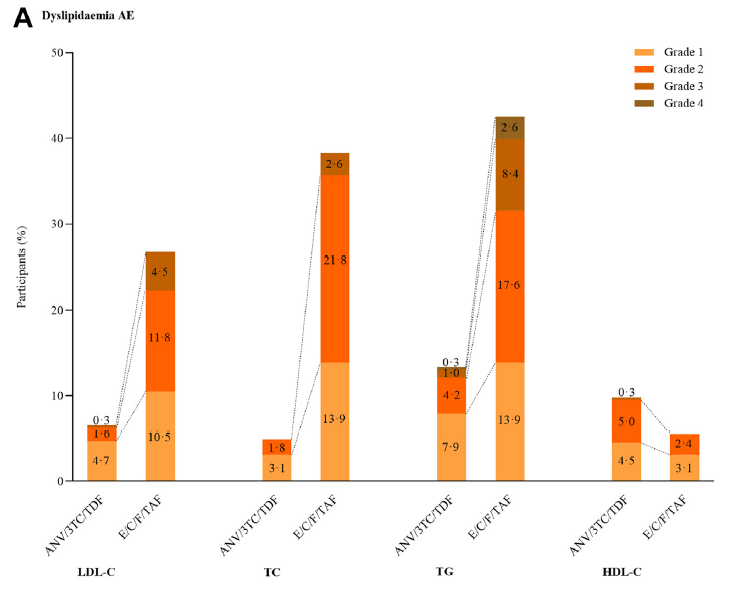
Proportion of Dyslipidemia-related Adverse Events by Severity
The long-term extension phases of the SPRINT study (96/144 weeks)[4-5] further confirmed sustained viral suppression efficacy and long-term tolerability of Aitribond®, supporting PWH in achieving their lifelong treatment goals.
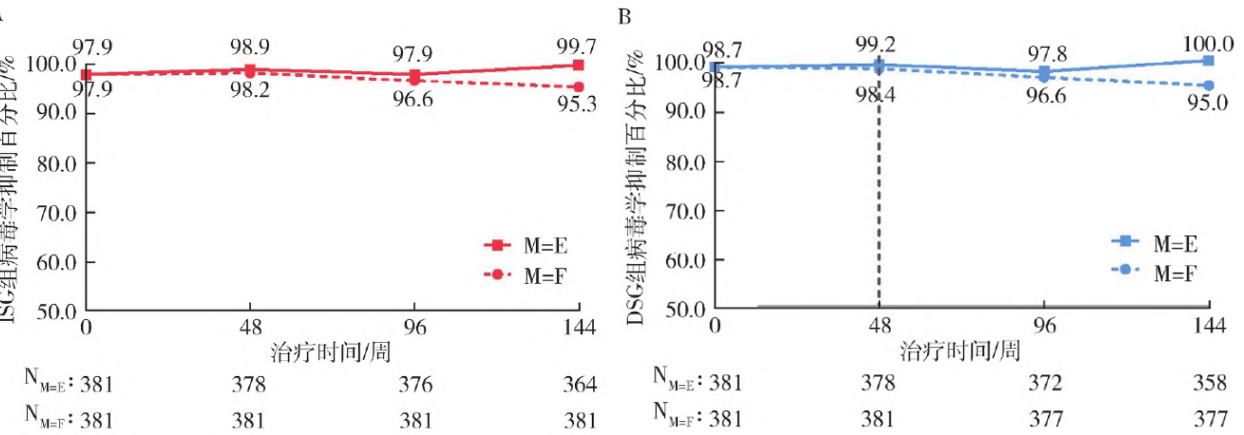
Virologic Suppression Percentage in ISG Group (A) and DSG Group (B) from Weeks 0 to 144
The citation of the Aitribond® series of studies in a Nature sub-journal is not only an honor but also a new starting point for responsibility and motivation. From clinical breakthroughs to international recognition, from drug approval to the accumulation of long-term evidence, Aitribond® - as a new generation of combination therapy for HIV - holds the potential to continue leading the current and future pathways of antiretroviral treatment, offering long-term treatment options for more patients.

[1] Tan R, Hua H, Zhou S, et al. Current landscape of innovative drug development and regulatory support in China. Sig Transduct Target Ther. 2025;10:220.
[2] Su B, Gao G, Wang M, et al. Efficacy and safety of ainuovirine versus efavirenz combination therapies with lamivudine/tenofovir disoproxil fumarate for medication of treatment-naïve HIV-1-positive adults: week 48 results of a randomized controlled phase 3 clinical trial followed by an open-label setting until week 96. Lancet Reg Health West Pac. 2023;36:100769.
[3] Zhang F, Wu H, Cai W, et al. Switch to fixed-dose ainuovirine, lamivudine, and tenofovir DF versus elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide in virologically suppressed people living with HIV-1: the 48-week results of the SPRINT trial, a multi-centre, randomised, double-blind, active-controlled, phase 3, non-inferiority trial. Lancet Reg Health West Pac. 2024;49:101143.
[4] Zhang F, Cai W, Wu H, et al. Immediate and delayed switches to tenofovir DF-containing, ainuovirine-based antiretroviral regimen: the SPRINT extensional study. BMC Med. 2025;23:524.
[5] 杨涤, 吴昊, 蔡卫平, 等. 基于真实世界数据的艾诺米替转换治疗HIV-1感染者144周有效性和安全性研究. 中国艾滋病性病. 2025;31(9):952-9.

